Dangerous pH Levels
Low or high pH solutions can be hazardous to different tissues. Eyes and mucous membranes are prone to damage by less extreme pHs, while skin can be irritated or burned by wider extremes of pH. Low pH can be dangerous due to acidity. High pH can be caustic and likewise cause tissue damage.
Acidity
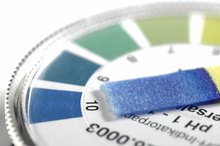
The measurement of pH is used to determine the concentration of acid (hydronium ion) in solutions. A pH of 7 is considered neutral, and represents the pH of pure water. A pH lower than 7 indicates an excess of hydronium ion (acid). Each digit represents a 10-fold increase in acid concentration, so a pH 6 solution is 10 times more acidic than pH 7. Acidic solutions tend to taste sour and do not feel slippery if rubbed between your finger and thumb.
- The measurement of pH is used to determine the concentration of acid (hydronium ion) in solutions.
Alkalinity
Diseases Caused by Alkaline or Acid pH in the Body
Learn More
A pH above 7 indicates an excess of hydroxyl ions. High pH solutions are called basic or alkaline. Excess alkalinity is caustic and can cause tissue damage. A pH of 8 is 10 times more basic than pH 7; pH 9 is 100 times more basic than pH 7. Basic solutions taste bitter and feel slippery when rubbed between your finger and thumb.
- A pH above 7 indicates an excess of hydroxyl ions.
- High pH solutions are called basic or alkaline.
The pH of Humans
Different fluids in the human body have different normal pH values 1. The normal pH of tears is about 7.1 or slightly higher. Saliva is slightly acidic at around 6.4. Sweat is also slightly acidic at between 4 and 6.8. Blood pH is normally slightly basic at 7.4. Stomach acid is less than pH 2.
- Different fluids in the human body have different normal pH values 1.
- Sweat is also slightly acidic at between 4 and 6.8.
Hazardous pH
How to Read Arterial Blood Gases
Learn More
When pH drops below about 2.5, it is considered potentially dangerous. It depends on what body tissues the low-pH liquid touches. While it is certainly safe to eat a lemon with pH 2.5, you don't want to get lemon juice in your eye. Prolonged exposure on the skin or mucous membranes will also cause irritation and tissue damage. Any liquid with a pH of 10 or more can cause burns depending on the tissue it touches and how long the tissue is exposed. Note that some liquids, such as milk of magnesia, are higher than pH 10 because they are meant to counteract stomach acidity. Take as directed.
- When pH drops below about 2.5, it is considered potentially dangerous.
- Any liquid with a pH of 10 or more can cause burns depending on the tissue it touches and how long the tissue is exposed.
Blood pH
Blood pH can drop in a condition known as acidosis. This can have several causes and can be treated with diet and medications. Alkalosis is a blood condition in which pH is elevated above normal. Blood pH levels are normally slightly basic, at 7.35 to 7.45, but become dangerously low with just a slight downward shift. The kidneys adjust blood pH under normal conditions.
- Blood pH can drop in a condition known as acidosis.
Related Articles
References
Writer Bio
This article was written by the CareerTrend team, copy edited and fact checked through a multi-point auditing system, in efforts to ensure our readers only receive the best information. To submit your questions or ideas, or to simply learn more about CareerTrend, contact us [here](http://careertrend.com/about-us).