What does fact checked mean?
At Healthfully, we strive to deliver objective content that is accurate and up-to-date. Our team periodically reviews articles in order to ensure content quality. The sources cited below consist of evidence from peer-reviewed journals, prominent medical organizations, academic associations, and government data.
The information contained on this site is for informational purposes only, and should not be used as a substitute for the advice of a professional health care provider. Please check with the appropriate physician regarding health questions and concerns. Although we strive to deliver accurate and up-to-date information, no guarantee to that effect is made.
How to Document a TB Test Reaction
A negative TB skin test is required for admission to a variety of institutions such as schools, nursing homes and assisted living facilities, as well as for employment purposes such as for teachers, day care or health care workers 1. Mycobacterium tuberculosis is extremely infectious and can spread rapidly. It is important for nurses and physicians to understand how to properly administer and read the results of a TB skin test to document the findings correctly 1.
If you are experiencing serious medical symptoms, seek emergency treatment immediately.
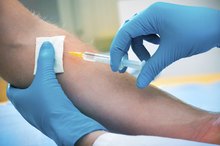
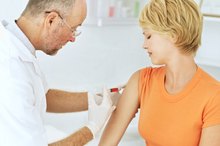
The PPD (purified protein derivative) test solution contains Mycobacterium tuberculosis antigens, not live bacteria. It is designed to produce a localized skin reaction in persons who may have TB. A small sample (0.1 ml) is injected directly under the first layer of skin on the forearm. Inserting the needle too deeply can affect the accuracy. A small bubble resembling a blister should result.
Prescreening clients is also key to accuracy. Factors such as recent exposure to TB, HIV status, drug use and any underlying immunosuppressive disorders such as cancer can affect results. Diabetes, kidney disease and previous inoculation with BCG vaccine may also alter the results. Recent exposure to live virus in other vaccines may also alter the results.
Signs of a Positive TB Test
Learn More
The TB skin test is left uncovered for 48 to 72 hours 1. The client should not disturb the site during this time. The site may itch and become reddened, but should not be rubbed or scratched.
The date, time and site of the TB skin test is documented on the report form 1. The Lot number, manufacturer and expiration date of the PPD solution should also be included.
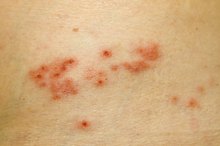
The injection site must be examined by a health care provider within 48 to 72 hours. The results are recorded on the screening form in millimeters of induration (redness and/or swelling) ranging from 0 mm upward.
Any redness is measured across the diameter. To be considered a positive reaction, the site must also be raised or swollen. The nurse or physician should feel the site as it may be difficult to visualize the raised area. The size of the raised area is documented as mm of induration. Any additional information such as recent exposure and symptoms such as coughing, sneezing, night sweats, should be documented.
What Is an SAP 10-50 Test?
Learn More
If the site is not raised, the result is considered negative. The size of any red area should be measured and recorded, but noted that no raised area is felt which determines the negative status. If the site is clear, the results are recorded as 0 mm.
According to the Centers and Disease Control and Prvention, induration of less than 10 mm is considered negative. However, the general health status of the client may change this. Anyone with a positive HIV status or altered immune status may be considered positive, after further evaluation, even if the induration is as low as 4mm. This information must be documented to support the exception.
Tips
A second skin test may be administered in one to two weeks to verify negative status. Anyone with positive results should be evaluated immediately for possible active or latent TB and treatment.
Related Articles
References
- Lab Tests Online; TB Skin Test
- CDC; Tuberculin Skin Testing
- Centers for Disease Control and Prevention. Testing for TB Infection. Updated April 14, 2016.
- University of Michigan. Michigan Medicine. Tuberculosis (TB). Updated July 30, 2018.
- Merck Manual, Consumer Version. Tuberculosis (TB). https://www.merckmanuals.com/home/infections/tuberculosis-and-leprosy/tuberculosis-tb#v785596
- U.S. Centers for Disease Control and Prevention (CDC). Fact Sheet: BCG Vaccine. Updated September 12, 2016
- U.S. Centers for Disease Control and Prevention (CDC). Fact Sheet: Tuberculin Skin Test. Updated September 8, 2016.
- University of Michigan. Michigan Medicine Health Library. Other Conditions With Similar Symptoms to Tuberculosis (TB).
Writer Bio
Kathy Quan RN BSN has been a nurse and a writer for more than 30 years. She is the author of four books including "The Everything New Nurse Book" and "The Everything Guide to Caring for Aging Parents." Quan writes for several websites including UltimateNurse.com and NurseTogether.com.