What does fact checked mean?
At Healthfully, we strive to deliver objective content that is accurate and up-to-date. Our team periodically reviews articles in order to ensure content quality. The sources cited below consist of evidence from peer-reviewed journals, prominent medical organizations, academic associations, and government data.
The information contained on this site is for informational purposes only, and should not be used as a substitute for the advice of a professional health care provider. Please check with the appropriate physician regarding health questions and concerns. Although we strive to deliver accurate and up-to-date information, no guarantee to that effect is made.
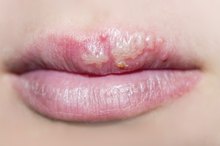
How to Get Rid of a Fever Blister on a Lip
Fever blisters, also known as cold sores, are caused by the herpes simplex 1 virus (HSV-1) 2. After the first infection and outbreak, which often occurs in childhood, this virus remains in the body, often in a dormant or inactive stage. However, HSV-1 can become active and cause repeated outbreaks during illness, stress, if your immune system is weak, or when the lip is exposed to trauma or excess sun. These blisters can be painful and embarrassing, and may seem to take forever to clear up. While there is no current way to get rid of HSV-1 in the body, there are steps you can take to lessen pain and discomfort, and reduce the number of days you experience an outbreak.
If you are experiencing serious medical symptoms, seek emergency treatment immediately.
Without treatment, most fever blisters take a few weeks to heal. Consider using an over-the-counter (OTC) product to speed up the healing process. Abreva (docosanol 10 percent cream) is a readily available OTC option that works by blocking the replication of the virus, however it needs to be applied at the first sign of a blister to be effected. When you feel that characteristic itching and burning that precedes the eruption of the blister, apply Abreva right away, then 5 times daily for up to 10 days.
Hydrogen Peroxide & Cold Sores
Learn More
For faster healing time, another option is to try Viroxin, an single-dose OTC product only available through dental professionals. A comparison of the effectiveness of Abreva and Viroxin was reported in the April 2011 issue of “Journal of Esthetic and Restorative Dentistry.” This study concluded that both OTC products worked to reduce healing time compared to no treatment 4. But on average, Viroxin users experienced healing in 4 days, compared to 7.6 days for Abreva and 11.5 days for no treatment. In addition, Viroxin users experienced faster pain relief.
Since antiviral agents are not likely to help once the lesion has erupted, sometimes the only available strategy is to relieve pain and prevent the spread of the infection. In this situation, try using lip balm containing cocoa butter or petroleum jelly in order to keep the lesion moist and prevent cracking, which increases the risk of other infections. According to a June 2013 review published in “U.S. Pharmacist,” once the lesion has already formed, a topical anesthetic can also be helpful. For example, Orabase, a 20 percent benzoyl peroxide ointment, can be applied to treat pain and form a barrier to shield the sore from irritation while it heals.
What Are the Dangers of Herpes if Not Treated?
Learn More
If you suffer from recurrent cold sores or fever blisters, talk to your doctor about prescription options. According to the article in U.S. Pharmacist, there are several antiviral medications that can be used, including acyclovir (Zovirax), famciclovir (Famvir) and valacyclovir (Valtrex). If taken at the first sign of a blister, these medications can reduce healing time and reduce the frequency of outbreaks, but they are not a cure for HSV-1. These medications can also have side effects, so your doctor will need to weigh the risks and benefits of antiviral therapy.
Fever blisters or cold sores are usually not serious. But if you have a compromised immune system, or if an infant is exposed to the HSV-1 virus, serious illness can result. Be sure to inform your doctor if you have sores that last more than 2 weeks, if your sores are accompanied by a fever, or if the sores are so severe it is difficult to swallow or eat.
Tips
If you are prone to fever blisters, wear a lip balm with sunscreen anytime you will be exposed to the sun for more than a few minutes. Replace your lip balm when the breakout is over to prevent the spread of bacteria.
Warnings
Fever blisters are contagious, so please keep your lips to yourself when you are experiencing an outbreak. Also, avoid touching the healing blister with your hands because you could spread the virus to other sites.
Reviewed by Kay Peck, MPH RD
Related Articles
References
- U.S. Pharmacist: Management of Common Oral Sores
- Merck Manual: Herpes Simplex Virus (HSV) Infections
- Cochrane Database of Systematic Reviews: Interventions for Prevention of Herpes Simplex Labialis (Cold Sores on the Lips)
- Journal of Esthetic and Restorative Dentistry: Treatment of Herpes Labialis: Comparison of Two OTC Drugs and Untreated Controls
- Ramchandani M, Kong M, Tronstein E, et al. Herpes Simplex Virus Type 1 Shedding in Tears and Nasal and Oral Mucosa of Healthy Adults. Sex Transm Dis. 2016;43(12):756-760. doi:10.1097/OLQ.0000000000000522
- Institute for Quality and Efficiency in Health Care (IQWiG). Cold sores: Overview. 2018.
- Montgomery-cranny JA, Wallace A, Rogers HJ, Hughes SC, Hegarty AM, Zaitoun H. Management of recurrent aphthous stomatitis in children. Dent Update. 2015;42(6):564-6, 569-72. doi:10.12968/denu.2015.42.6.564
- Krissel JD, Bhatia A, and Thomas A. Cold sore susceptibility gene-1 genotypes affect the expression of herpes labialis in unrelated human subjects. Hum Genome Var. 2014; 1: 14024. doi:10.1038/hgv.2014.24
- Fatahzadeh M and Schwartz RA. Human herpes simplex virus infections: Epidemiology, pathogenesis, symptomatology, diagnosis, and management. J Am Acad Dermatol. 2007;57(5):737-63. doi:10.1016/j.jaad.2007.06.027
- Formica M, Kabbara K, Clark R, Mcalindon T. Can clinical trials requiring frequent participant contact be conducted over the Internet? Results from an online randomized controlled trial evaluating a topical ointment for herpes labialis. J Med Internet Res. 2004;6(1):e6.
- American Academy of Dermatologists. Cold sores: Overview.
- American Academy of Dermatologists. Cold sores: Signs and symptoms.
- American Academy of Dermatologists. Cold sores: Tips for managing.
Resources
Tips
- Wear a balm containing sunscreen on your lips anytime you will be exposed to the sun for more than a few minutes if you are prone to fever blisters.
- Chocolate, peanuts or almonds may increase irritation, so avoid these foods if you are having a fever blister outbreak or feel one coming on.
Warnings
- Replace your lip balm or cut off at least ¼ inch when the breakout is over to prevent the spread of bacteria.
- Fever blisters are contagious, so please keep your lips to yourself when you are experiencing an outbreak.
- Avoid touching the healing blister with your hands because you could spread the virus to other sites.
Writer Bio
Jamie Simpson is a researcher and journalist based in Indianapolis with more than 10 years of professional writing experience. She earned her B.S. in animal science from Purdue University and her Master of Public Affairs in public management from Indiana University. Simpson also works as a massage therapist and equine sports massage therapist.